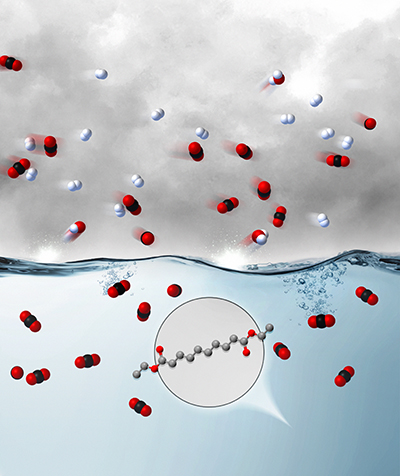
 The invention is a family of hydrophobic, low viscosity, low vapor pressure physical solvents with molecular structures consisting of two or more alkyl-ester functional groups on a central hydrocarbon chain. These solvents have been shown to possess high carbon dioxide (CO2) solubility and absorption selectivity, which make them well suited for the removal of CO2 from hydrogen (H2) produced from synthesis gas. This technology is available for licensing and/or further collaborative research from the U.S. Department of Energy’s National Energy Technology Laboratory.
The invention is a family of hydrophobic, low viscosity, low vapor pressure physical solvents with molecular structures consisting of two or more alkyl-ester functional groups on a central hydrocarbon chain. These solvents have been shown to possess high carbon dioxide (CO2) solubility and absorption selectivity, which make them well suited for the removal of CO2 from hydrogen (H2) produced from synthesis gas. This technology is available for licensing and/or further collaborative research from the U.S. Department of Energy’s National Energy Technology Laboratory.
Challenge
Future integrated gasification combined cycle (IGCC) power plants and steam methane reforming (SMR) chemical plants have the potential to reduce the cost of CO2 capture. These power and chemical plants generate high-pressure CO2 gas streams from the in-situ water gas shift reaction when producing H2 used to power the electrical turbines. A variety of methods have been proposed to capture CO2, including solvent, sorbent, and membrane technologies, with continuous solvent looping systems currently considered to be the most advanced. Precombustion capture of CO2 is typically accomplished using physical solvents.
State-of-the-art precombustion CO2 capture processes predominantly employ hydrophilic physical solvents. Current commercial physical solvents touted for IGCC CO2 capture were developed for removing acid gases from raw natural gas streams. Therefore, they were designed to remove significant amounts of water from the process gas. As such, the focus was on the purification of the process gas with less concern for generation of high-purity CO2 streams suitable for pipeline transmission and sequestration. While water removal is important for natural gas pipeline applications, it is not favorable for applications in which the fuel stream is directly combusted on-site, as would be encountered in IGCC systems.




 The invention is a family of hydrophobic, low viscosity, low vapor pressure physical solvents with molecular structures consisting of two or more alkyl-ester functional groups on a central hydrocarbon chain. These solvents have been shown to possess high carbon dioxide (CO2) solubility and absorption selectivity, which make them well suited for the removal of CO2 from hydrogen (H2) produced from synthesis gas. This technology is available for licensing and/or further collaborative research from the U.S. Department of Energy’s National Energy Technology Laboratory.
The invention is a family of hydrophobic, low viscosity, low vapor pressure physical solvents with molecular structures consisting of two or more alkyl-ester functional groups on a central hydrocarbon chain. These solvents have been shown to possess high carbon dioxide (CO2) solubility and absorption selectivity, which make them well suited for the removal of CO2 from hydrogen (H2) produced from synthesis gas. This technology is available for licensing and/or further collaborative research from the U.S. Department of Energy’s National Energy Technology Laboratory.