The Technology
Direct Air Capture (DAC) for the removal of atmospheric CO2 is a promising technology for the global transition to net zero carbon emissions. Solid adsorbents are the current material of choice for DAC because of their high porosity, relatively fast kinetics, and low-cost regeneration compared to solvent-based CO2 capture technologies. Of the solid adsorbents, porous carbon represents one of the better options due to its low cost and scalable preparation. However, this material displays poor selectivity for low concentrations of CO2. It also requires a high adsorption temperature and thus, significant energy for regeneration.
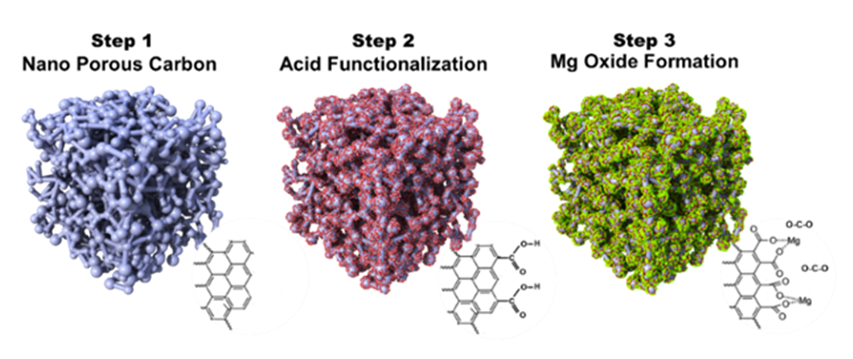
NETL has developed a cost-effective metal oxide functionalized carbon sorbent that displays high selectivity to CO2 in ambient conditions and requires low adsorption temperature. Synthesis is achieved via a three-step process. First, porous coal or any other porous carbon source is thermally decomposed under basic conditions, converting it to a highly nanoporous carbon (NPC) sorbent with high surface area. The NPC is then oxidized by acid groups (-COOH) to afford NPC-acid. Finally, metallic ions, such as magnesium (Mg+2), are tethered to the acid functional groups to generate metal oxide moieties throughout the highly porous scaffold.
This magnesium oxide (MgO)-functionalized nanoporous carbon sorbent (MgO-NPC) exhibits high CO2 selectivity at four times greater than that of a non-MgO functionalized NPC, and comparable to that of the best performing conventional functionalized porous carbon sorbents for DAC. Further, the MgO-NPC sorbent has a high affinity for CO2 in ambient conditions, displaying CO2 uptake 50 times greater than an unmodified NPC. This high performance can be maintained over multiple adsorption/desorption cycles. Lastly, sorbent regeneration is achieved at a low temperature compared to similar materials, translating into significant energy savings.